Landmark ALL-RISE Trial results
and simultaneously published in the
CathWorks FFRangio Demonstrated Similar Clinical Outcomes to Pressure Wire-guided Treatment
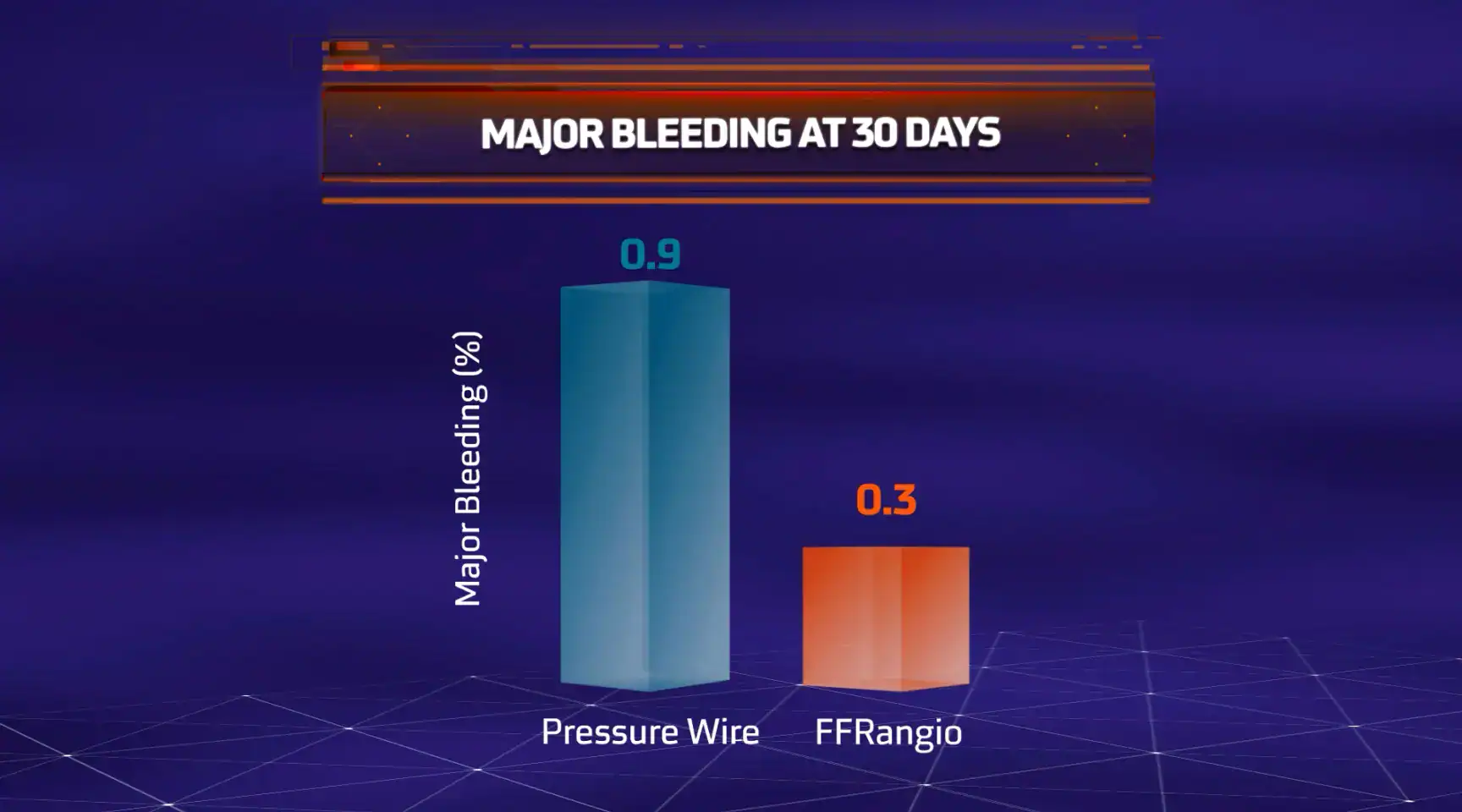
CathWorks FFRangio is the only FDA-approved angiography-derived physiology (ADP) technology proven to FFR/NHPR in MACE* outcomes
*
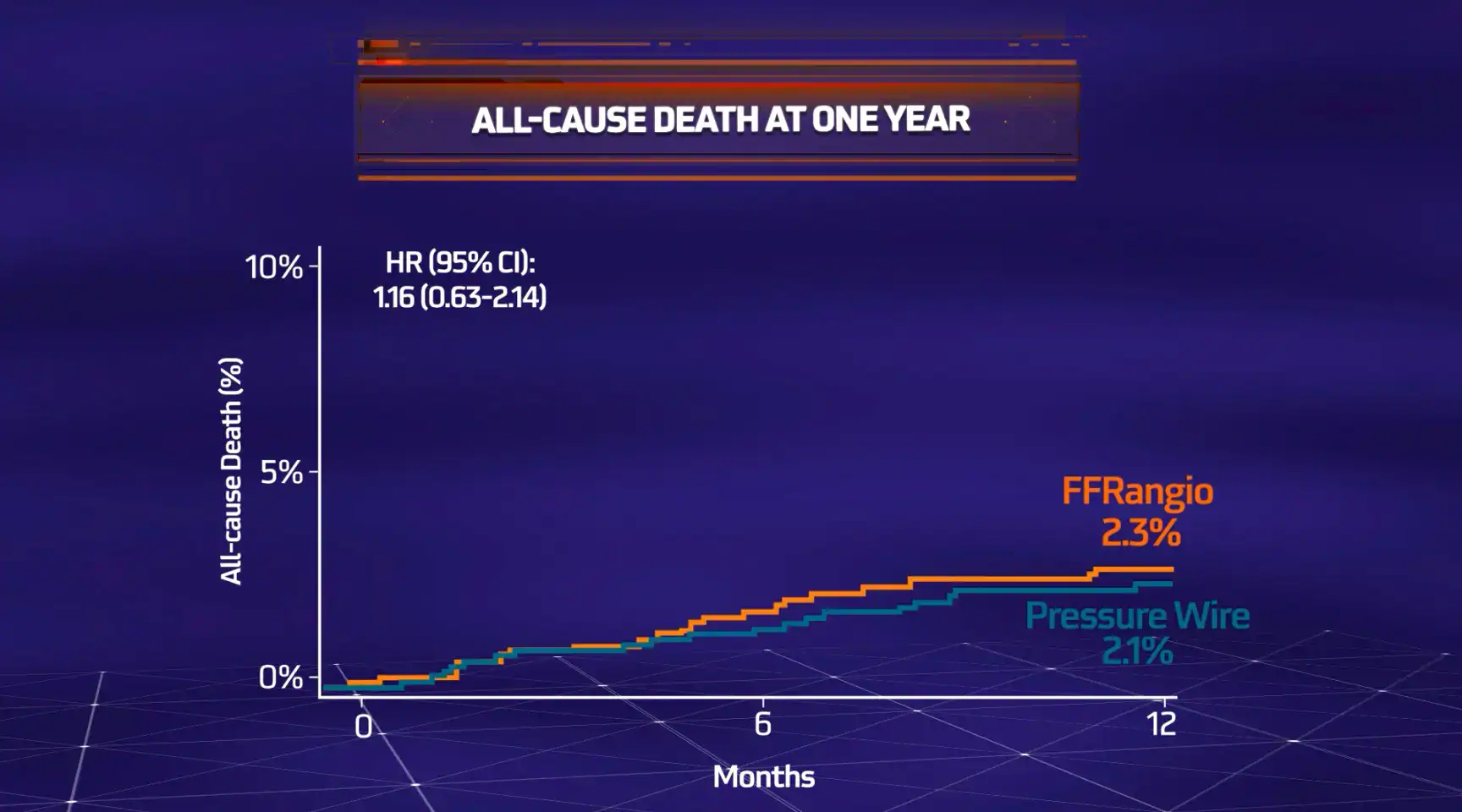
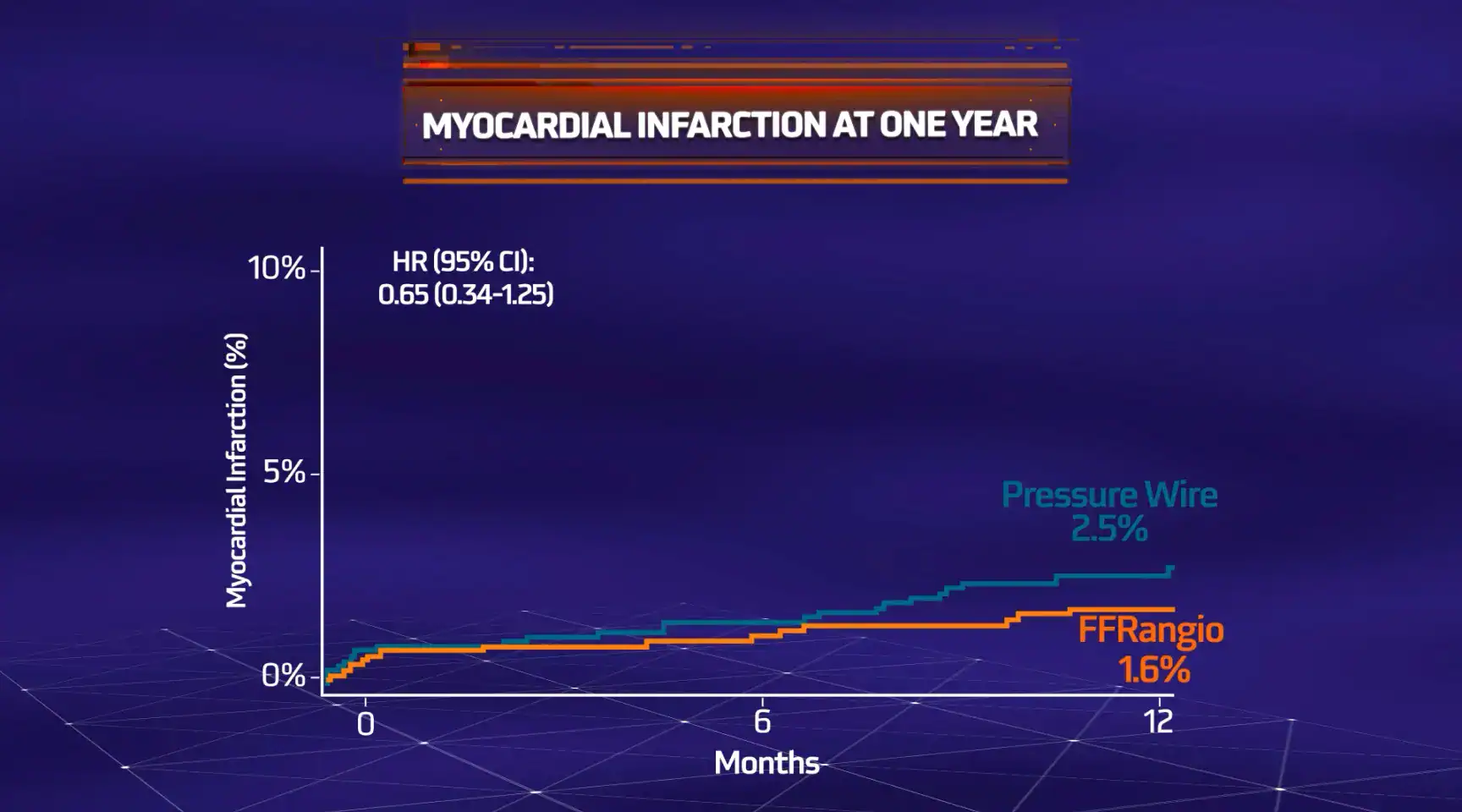
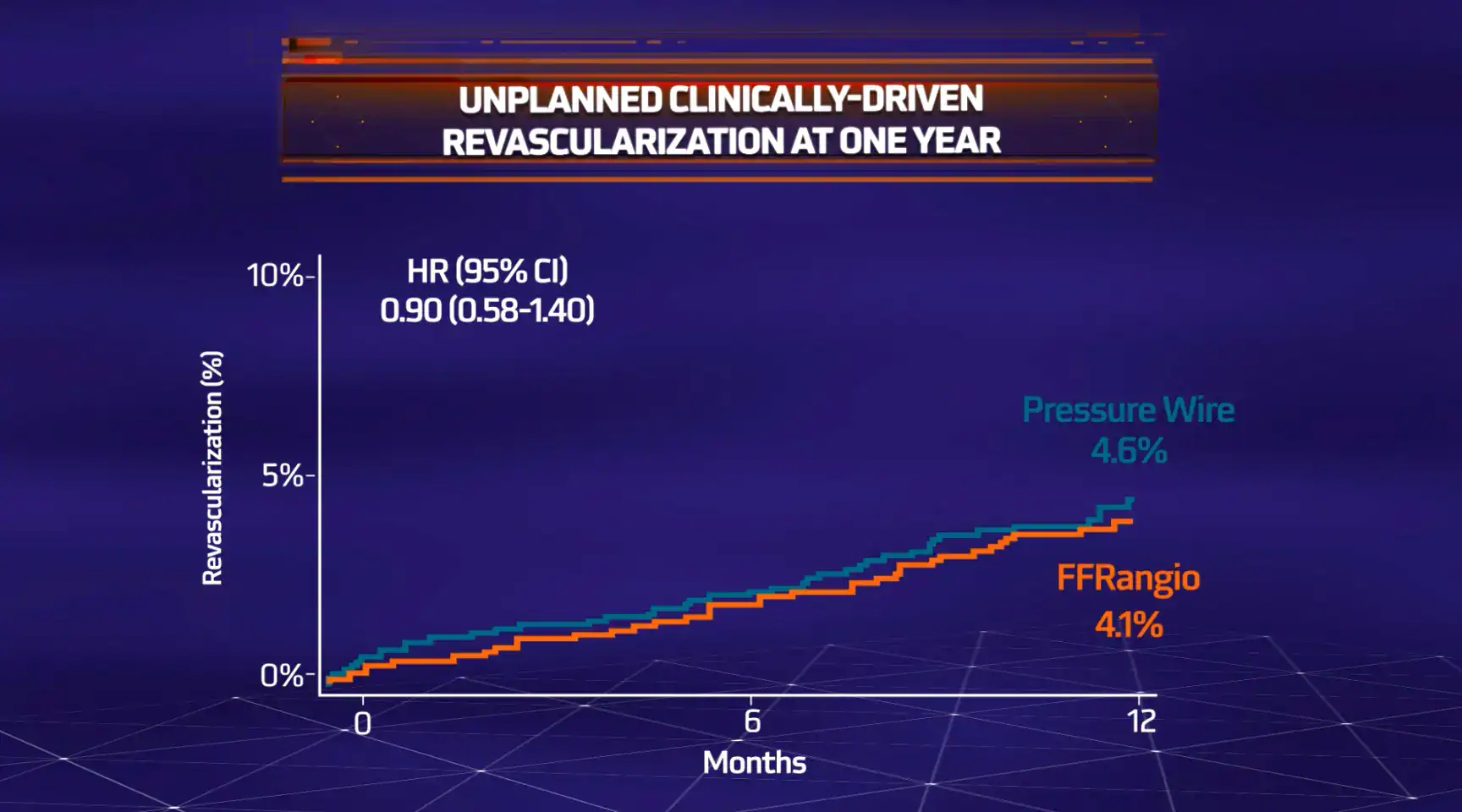
MACE = All-cause death, myocardial infarction (MI) or unplanned clinically-driven revascularization
CathWorks FFRangio:
The New Standard of Care in Coronary Physiology
Study Objective
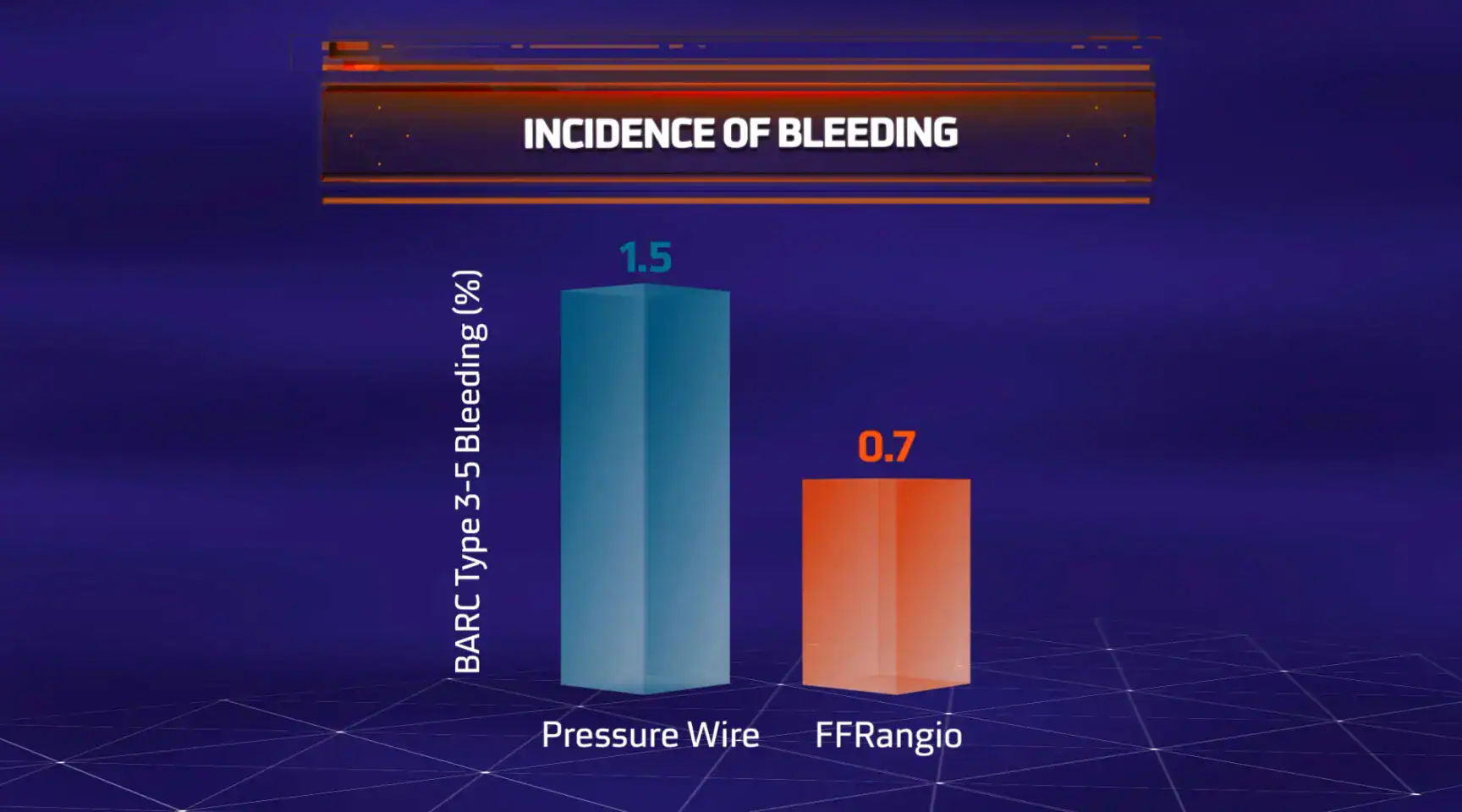
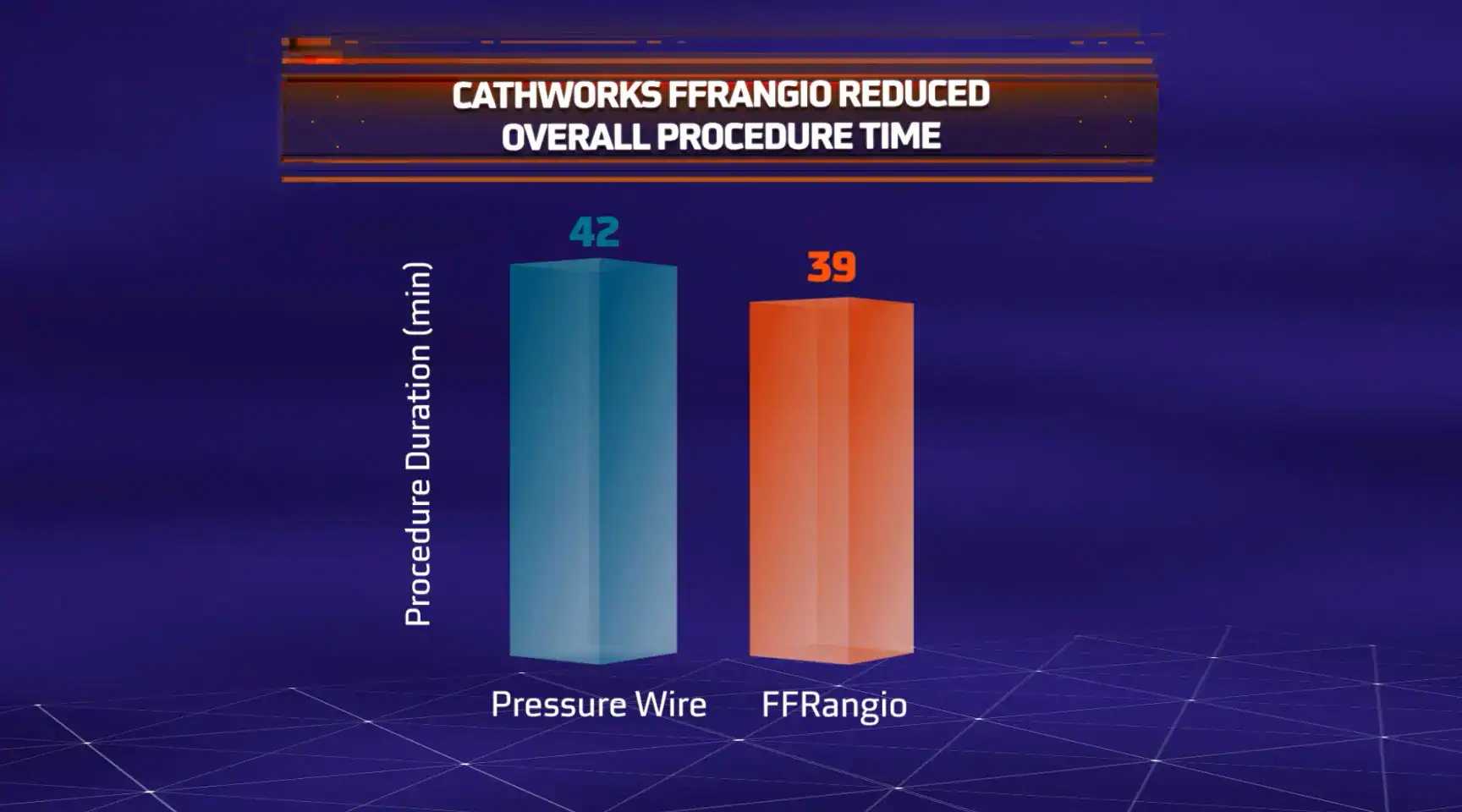
The objective of the ALL-RISE Trial was to assess whether CathWorks FFRangio is comparable to conventional pressure wire-based (FFR/NHPR) assessment of intermediate lesions being considered
Study Design
Patients presenting with coronary stenoses of intermediate significance were randomized to physiological assessment with CathWorks FFRangio (FFRangio group; 965 patients) or an invasive pressure wire-based approach (pressure wire group; 965 patients). Participating sites could choose whether the patients randomized to the pressure wire group were assessed with fractional flow reserve (FFR), requiring drug stimulation, or non-hyperemic pressure

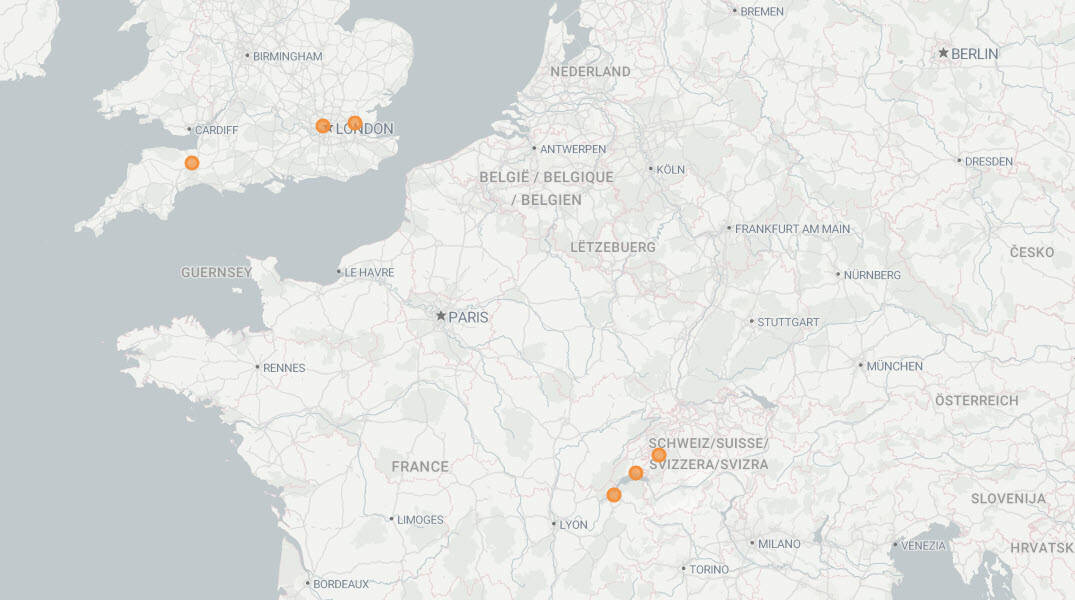
Participating Sites
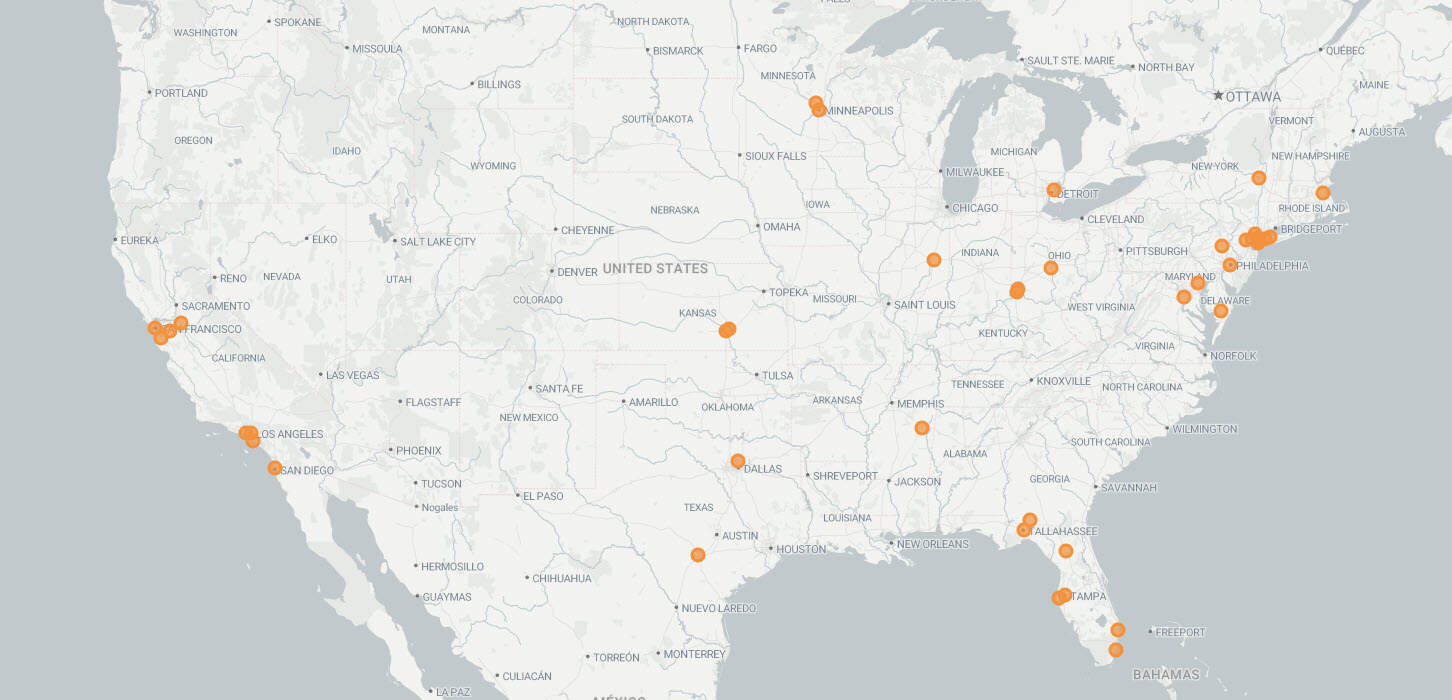
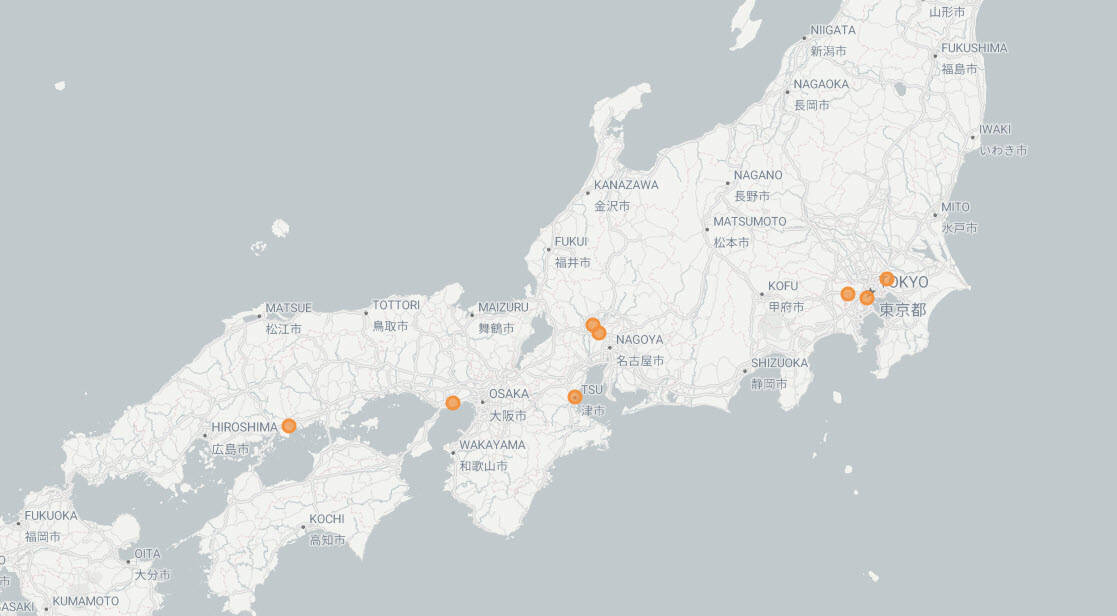
The ALL-RISE Study included 59 sites across North America, Asia, Europe and the
1.
Kirtane AJ et al. A large-scale, global randomized trial of coronary physiology derived from conventional angiography compared with an invasive pressure wire-based approach to guide percutaneous coronary intervention, as presented at ACC 2026. ClinicalTrials.gov number NCT05893498.
2.
Fearon WF et al. Angiography-derived fractional flow reserve to guide PCI. N Engl J Med, published on March 29, 2026. DOI: 10.1056/NEJMoa2600949.