FFRangio Demonstrated Similar Outcomes to FFR in PROVISION Trial
2025
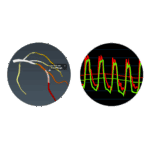

FFRangio Demonstrated Similar Outcomes to FFR in PROVISION Trial

ALL-RISE Global Study Completed Enrollment of 1,927 Patients

FFRangio System Received EU‑MDR Approval

First Patient Enrolled in ALL-RISE Global Clinical Study

Co-Promotion Agreement Announced

FFRangio System Realworld Outcomes Study Published

4th Generation of FFRangio System Launched

FFRangio System Received Japan PMDA Approval

FFRangio System Received US FDA 510(k) Market Clearance
FFRangio System Received CE Mark

CathWorks Founded

Ramin Mousavi brings extensive executive leadership and operational expertise across various domains including general management, marketing, strategy, product development, and commercialization within diverse market segments. With a notable tenure most recently leading the patient monitoring and digital health portfolio at Baxter International, Ramin has also held key leadership roles at Edwards Lifesciences, Panasonic, and Rockwell Collins.
At CathWorks, Ramin served as Vice President of Global Marketing and Strategy, as well as Chief Marketing Officer from 2019 to 2020. He holds dual B.S. degrees in Computer and Electrical Engineering from the University of California, Irvine, complemented by an M.B.A. from the Paul Merage School of Business at UC Irvine. Additionally, Ramin has furthered his expertise with a Healthcare Executive Leadership certificate for Business Innovation in Global Healthcare from Harvard Business School.

Dalit Cohen-Shipperman brings over 14 years of executive-level experience in Human Resources to CathWorks. Throughout her career, she has spearheaded global HR processes across diverse organizations, contributing to both tactical and strategic growth initiatives. With a focus on high-tech sectors including telecom, communications intelligence, and network security, Dalit has played a pivotal role in driving organizational success.
Dalit holds a B.A. in Psychology and Communications from Haifa University, and she earned her M.A. cum laude in Organizational Psychology from Bar-Ilan University. Her academic background enriches her understanding of human behavior within the workplace, allowing her to implement effective HR strategies that align with the company’s objectives.
Beyond her professional endeavors, Dalit enjoys a variety of activities outside of work, including ballet dancing, running, reading, and hiking, showcasing her well-rounded interests and commitment to a balanced lifestyle.

Mike Feher brings 15 years of seasoned leadership and operational expertise to CathWorks, with a focus on financial planning and analysis, process improvement, resource allocation, and investor relations. His background includes spearheading strategic financial initiatives at Allied Universal, where he played a pivotal role in driving revenue growth, cost reduction, and international market expansion.
During his tenure at Edwards Lifesciences, Mike held diverse leadership positions spanning business finance, corporate strategy, and investor relations, contributing significantly to the company’s success and growth trajectory.
Mike holds a B.S. degree in Accounting and Business Administration, specializing in Finance and Economics, from Chapman University, and is a licensed CPA. Additionally, he earned an M.B.A. in Corporate Finance from the Marshall School of Business at the University of Southern California, further solidifying his expertise in financial management and strategy.
As a native of Southern California, Mike cherishes spending time with his family, enjoying the local beaches and pursuing his passion for surfing whenever possible.

Asaf Jazcilevich brings a wealth of experience in R&D management to CathWorks, with a strong record of success in building and leading multi-disciplinary teams encompassing software, hardware, and algorithm engineering across diverse sectors including mobile, display, and automotive technologies.
With over 12 years of senior leadership at Samsung’s Israeli R&D Center, Asaf spearheaded the development of cutting-edge computer vision and image processing platforms, which were subsequently integrated into Samsung’s flagship products. Prior to his tenure at Samsung, he served as a firmware team leader at TransChip and gained valuable experience at Intel.
Asaf holds a B.Sc. in Electrical Engineering from Tel-Aviv University, underscoring his technical expertise and academic foundation. Beyond his professional endeavors, he enjoys a variety of hobbies including running, working out, playing the cello, and solving word games.

Sarita Monico joins CathWorks with over two decades of medical device strategic marketing experience. Sarita was integral to the launch and commercialization of the revolutionary transcatheter aortic valve replacement therapy and multiple other lifesaving innovations. Most recently, Sarita led the U.S. commercial marketing efforts for the industry-leading electrophysiology portfolio at Johnson & Johnson. She earned her B.S. in Biochemistry and Cell Biology from UC San Diego.
Outside of work, Sarita enjoys spending quality time with her daughter on the tennis court, volunteering and traveling.

Fabian Ngo brings nearly 20 years of cross-functional expertise to his role at CathWorks, bringing in experience from his roles in human capital consulting, sales operations, market analytics, and product management. Fabian’s professional journey at Edwards Lifesciences has included pivotal roles and key contributions, such as supporting commercial operations for multiple generations of the TAVR platform, establishing the first advanced analytics team to understand clinician and patient dynamics, and leading the commercial strategy for the development of the next-generation monitoring platform.
Fabian earned his B.A. in Economics from Claremont McKenna College and an M.B.A. with a focus on Strategic Management and Marketing-Operations from the Wharton School of the University of Pennsylvania. Outside the office, Fabian is either on a bike trail, hiking up a mountain, or setting up camp in the woods. When not out exploring nature, he’s exploring the music scene, on the lookout for innovative upcoming artists.

Ayelet Peer brings over 15 years of extensive operational and executive experience in quality and regulatory affairs within the medical device and pharmaceutical industries to CathWorks. With a proven track record, she has effectively led regulatory audits and served as the primary regulatory liaison for communications and submissions to EU competent authorities, the U.S. FDA, and Health Canada.
Prior to joining CathWorks, Ayelet held leadership roles in Quality and Regulatory Affairs at S.L.P. and gained eight years of invaluable experience in various Quality and Regulatory Compliance positions at Teva.
She holds a B.S. in Biology and Chemistry, as well as an M.B.A. with a specialization in Entrepreneurship and Innovation Management, both from Tel-Aviv University, reflecting her dedication to both scientific rigor and strategic business acumen.
Outside of her professional pursuits, Ayelet enjoys indulging her creative side through painting and exploring the great outdoors through hiking.

Natalie Sickler brings a decade of experience in the meetings and events industry, running large scale trade shows, sales meetings and special customer events as part of the marketing teams of medical device companies such as Biosense Webster and Allergan Pharmaceuticals.
As Chief of Staff at CathWorks, Natalie plays a vital role in the planning and coordination of account launches, customer contract execution and management of overall co-promotion activities with Medtronic. When not working, Natalie enjoys traveling with friends and family, trying any new highly rated restaurants and weekend trips to the California and Oregon mountains to snowboard when the weather permits. She holds a B.S. from San Diego State University and completed her study abroad program at Oxford University.

Masashi Yamamoto brings extensive experience as a business executive in the medical devices industry in Japan to CathWorks. Beginning his journey with CathWorks as a consultant in February 2018, Masashi played a pivotal role in securing the PMDA’s approval of the FFRangio System in Japan. With the establishment of CathWorks Japan GK in early 2019, he assumed the role of General Manager, overseeing operations in the region.
Prior to joining CathWorks, Masashi held senior leadership positions at Japanese subsidiaries of renowned medical devices companies including Boston Scientific, Covidien, Draeger Medical, and Corin Group, accumulating over 30 years of management experience. Throughout his career, he has led the successful launches of innovative medical devices such as coronary stents, rotational atherectomy devices, intravascular ultrasound imaging devices, and other cutting-edge technologies in the Japanese market.
Masashi holds a B.Sci in Electronics and an MBA, further enriching his comprehensive skill set and business acumen. Outside of his professional pursuits, he finds solace in classical music, particularly German romantic repertoire, and enjoys attending orchestra concerts or opera performances whenever his schedule permits.